Induced pluripotent derived stem cell-derived OPCs
Derivation of OPCs
hES cell therapy suffers regulatory setbacks in the United States federal research funding scheme and is also not patient specific. This implies that any treatment using hES-derivatives would require the patient to be immune suppressed for an extended duration of time. Induced pluripotent stem (iPS) cells are fully reprogrammed cells which can be generated from a patient's fully differentiated, somatic cells.
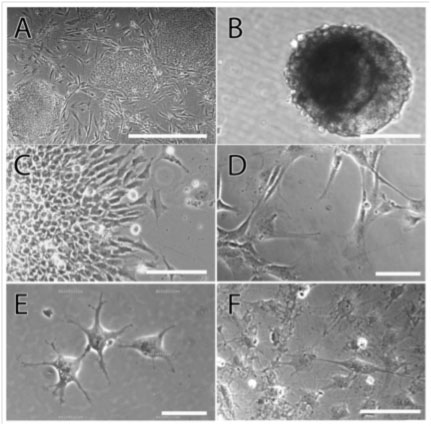
Figure 1
Figure 1: Bright field microscopy images of the various stages of OP differentiation from induced pluripotent stem cells of the BC1 line. A. The undifferentiated cells grew in colonies on a mouse embryonic fibroblast feeder layer, B. EBs were formed by suspending the undifferentiating cells in culture on ultra-low adherence plates, C. 15-day EBs were plated on matrigel, leading to the extension of cells of neuroectodermal lineage from the plated EB. Upon passaging, these neural progenitors grew in a monolayer and were subsequently differentiated to the glial progenitor (D) stage. E. Addition of PDGF-AA to the media led to GPs transitioning into the OP stage, displaying a rounded morphology with multiple filopodial extensions. F. Terminal differentiation was induced by the removal of PDGF-AA, leading to the appearance of mature oligodendrocytes displaying characteristic branching. Scale bars- A: 300 µm, B-F: 100 µm
Development

Figure 2
Figure 2. Plots showing mRNA expression of various developmental markers for all three iPS markers during various points in the OP differentiation timeline, as measured using qRT-PCR. Expression of pluripotency associated genes included A. OCT4, B. SOX2 and C. NANOG. Markers indicative of the oligodendrocyte lineage included D. Ng2, E. GALC, F. Mag, and G. Mobp. The bars represent results for the positive control, total RNA from human fetal brain.
Three iPS cell lines were chosen: BC1, MR31 and A1-4. Two of these (A1-4, MR31) were created using viral integration methods while the third (BC1) was created using a nonviral, non-integrative, episomal plasmid-based approach. This distinction was made because of greater clinical relevance of non-integrative iPS and also to ascertain which lines could generate OPCs more efficiently.
A protocol similar to that used for hES was used to derive OPCs from all three iPS lines. Immunocytochemistry confirmed expression of stage-specific markers and morphological appearance of each stage of cells was as expected.
RT-PCR analysis was used to quantify the expression of stage-specific and myelin-specific cellular markers and compare them across lines. In the retrovirally derived line (MR31), OCT4 levels remained upregulated compared to the other two lines. The non-integrative line, BC1, showed earlier expression of the myelin associated glycoprotein (MAG).
OPCs derived from the BC1 line were transplanted in rat spinal cords twenty four hours after moderate contusive SCI.

Figure 3
Figure 3: Indirect immunofluorescence detection defining the various states of IPS differentiation into OPCs (cells from BC1 iPS). The differentiated state and marker analyzed is indicated for A.- C. NPs; D.- F. GPs and G. -O. OPCs. Nuclei were stained blue using 4',6-diamidino-2-phenylindole (DAPI). P.-R. Quantification of the indirect immunofluorescence analyses calculating the percent of cells expressing a particular marker. P. 90% of all cells from each line express NPs early neural markers nestin and A2B5. Q. Likewise the number of GPs expressing markers of this stage NG2 and PDGFR were also similar among lines. R. In contrast, a higher percent of cells derived from BC1 expressed later OP markers MAG, MOBP and O1 while fewer cells from BC1 expressed early NP marker Nestin and early OP marker O4 compared to MR31 and A1-4. Asterisks denote statistical significance compared to BC1 (N = 9, P<0.05).
Myelination

Figure 4
Figure 4: Characterization of oligodendrocyte progenitors (OP) derived from the BC1 iPS line, transplanted 24 hours after a 12.5 mm contusion in rat spinal cord. iPS-derived OPCs transplanted 24 hours after injury survived up to 65 days following injection and differentiated to mature oligodendrocytes. A. Human specific nuclear staining (blue) showed that cells transplanted at the site of injury expressed myelin basic protein (MBP, red) and not the astrocyte associated marker GFAP (green). A few human cells migrated away from the site of injection and integrated into the intact host tissue; these are indicated by arrows. B. Shows staining of transplanted cells injected peripheral to the site of injury also express MBP. Dapi stains rat and human nuclei blue. C. Double stain of transplanted cells that express another marker of mature oligodendrocytes, myelin associated glycoprotein (MAG, green) but did not express a marker of the hematopoietic lineage D. CD45 (red). This is consistent with double immunostaining in sections with E.) MBP and F. PECAM, a marker of macrophages and T cells. Few CD45 or PECAM positive cells were detected at the site of cell injection indicating healthy engraftment of the transplanted OPCs without macrophage engulfment approximately two months after injury. B-F. Dapi stains rat and human nuclei blue. Tissue was isolated 65 days after cell injection in cyclosporin-treated rats. Asterisk denotes center of cell injection in all panels. Scale bar: 100 µm.

Figure 5
Figure 5: H&E and Luxol Fast Blue staining. Hematoxylin and eosin stained paraffin sections of spinal cords from animals subjected to contusive SCI and treated with iPS-derived OPs or heat-killed control cells. A. Heat-killed cells, 38 d. B. BC1-OPs, 30d. As expected for a 12.5 mm contusive injury, cavity formation was seen in both groups; however, the cavity manifests differently in the treated group (B), compared to the control group (A). The control shows more pronounced cavitation, which extends further from the epicenter along the central canal. This indicates that the transplantation of live OPs into the newly formed cavity 24 hours after injury limited the normal expected progression of this level of contusive injury. The asterisks indicate the center of the injury site. C, D. Luxol Fast Blue (LFB) staining of sagittal spinal cord sections showed significant reduction in staining in the control group, which received transplant of heat-killed BC1-OPs (C) as compared to the spinal cords of animals transplanted with BC1 iPS-derived OPs (D). Higher levels of LFB in the OPC treated rats C indicate the presence of greater amounts of myelin. The representative spinal cords shown in C and D were harvested 38 and 48 days after transplant respectively. C. The arrows point to cells which demonstrated intense cytoplasmic LFB staining, indicative of immune cell clearance and demyelination. W: white matter, G: grey matter.

Figure 6
Figure 6: Transmission electron microscopy shows increased post-injury remyelination in BC1 iPS-OP treated animals when compared to the heat-killed cell treatment control. A.-C. Transmission electron microscopy of sagittal sections of the spinal cord showed remyelination of spared axons by BC1-derived OP cells after contusive SCI in rats. A. Normal, endogenous myelination of an uninjured rat spinal cord as indicated by white arrows, showing thick and tightly-packed myelin sheaths. B. Remyelination observed in rats transplanted with BC1-derived OPs, seen 57 days after transplantation. The relatively thinner and loosely packed myelin sheaths may be compared with the normal condition shown in A, indicating that this is remyelination by the human cells. C. Multiple nude axons seen in a rat injected with heat-killed BC1-OPs, with insignificant remyelination, 57 days after transplant. Magnification (A-C): 10000X.








